[ad_1]
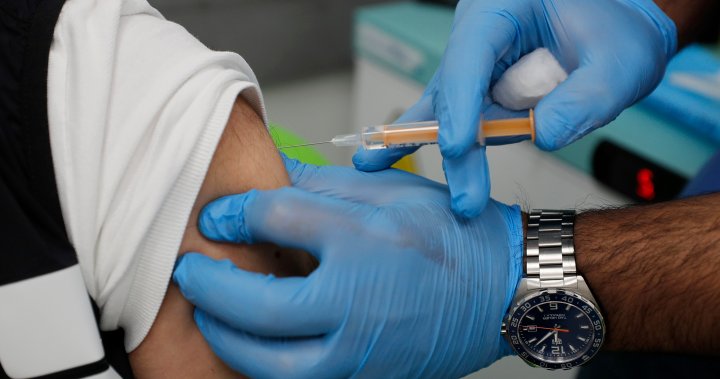
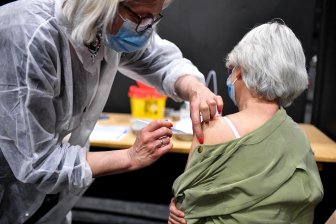
Britain’s medicines regulator said it has identified 30 cases of rare blood clot events associated with the AstraZeneca COVID-19 vaccine but stressed the benefits “continue to outweigh any risks.”
The Medicines and Healthcare Regulatory Agency said the risk associated with this type of blood clot is “very small” and that the public should continue to take up the vaccine when offered it.
The agency said late Friday the cases relate to the period up to March 24, during which 18.1 million doses of the vaccine had been administered and that it hadn’t received any similar reports with regard to the Pfizer-BioNTech vaccine.
Concerns over the AstraZeneca vaccine prompted some countries including Canada, France, Germany and the Netherlands to restrict its use to older people.

The World Health Organization has urged countries to continue using the jab.
© 2021 The Canadian Press
[ad_2]