[ad_1]
Topline
Canada’s health agency approved Pfizer’s Covid-19 vaccine for use in adolescents aged 12 to 15 Wednesday morning, Health Canada’s chief medical adviser announced, with the U.S. Food and Drug Administration set to approve the vaccine for all teenagers in the U.S.
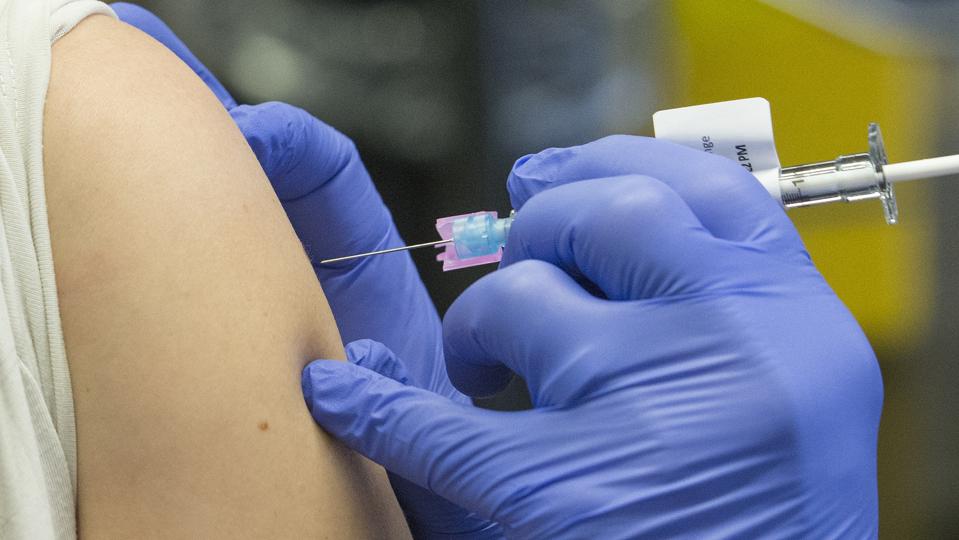
TORONTO, ON – APRIL 10: Dr. Michael Anderson (right) administers a dose of the Pfizer vaccine into … [+]
Toronto Star via Getty Images
Key Facts
Health Canada chief medical adviser Dr. Supriya Sharma said the announcement “marks a significant milestone in Canada’s fight against the pandemic.”
Fabien Paquette, the vaccine lead for Pfizer in Canada, said the “significant step forward” will help “protect adolescents before the start of the next school year.”
Health Canada began its review of Pfizer’s vaccine for adolescents on April 16.
Pfizer released data from a U.S. study of over 2,200 12- to 15-year-olds in March that showed there were no cases of Covid-19 in fully vaccinated volunteers.
Canada approved the Pfizer vaccine for use in December.
Key Background
Canada has relied on the Pfizer and Moderna vaccines in its vaccination effort. The country’s National Advisory Council on Immunization said Monday “the mRNA vaccines are the preferred vaccine.” Pfizer is set this week to double the number of vaccine doses it sends to Canada to 2 million a week. Roughly 34.5% of Canadians have received at least one dose of a Covid-19 vaccine.
Tangent
President Joe Biden announced Tuesday the U.S. will be able to make “20,000 pharmacy sites” available to distribute the Pfizer vaccine to adolescents between the ages of 12 and 15 “immediately” when the FDA approves the lowered eligibility. Biden cautioned “the FDA, and the FDA alone, will make that decision” but on Wednesday Fauci seemed sure the announcement was imminent. Fauci said he thinks “it’s going to be very soon” and he “cannot imagine it’s going to be much longer” than several days.
Further Reading
FDA To Expand Pfizer Covid-19 Vaccine Eligibility To 12-15-Year-Olds (Forbes)
Pfizer Will File For Full FDA Approval By End Of May, Expects $26 Billion In Vaccine Revenue (Forbes)
[ad_2]